DNA

I'M HIGHLY SUSPICIOUS of the growing obsession with genetic ancestry tests. 23andMe. AncestryDNA. People can now scrape their inner cheek with a swab, mail it to a company for $99, and brag to you about a cultural or racial epiphany they’ve had based on being 4.7 percent of something. Who knows how this personal genetic information might be used. I suspect these companies respect people’s privacy as much as Facebook does. I’ve heard that governments and police departments are already using this information to track people.
And yet, I would be lying if I said I haven’t thought about purchasing a DNA kit for myself. Yes, I know that such tests provide limited and potentially misleading information. Yes, I understand that they fuel problematic framings of race that tie race to genetics when race is actually something socially and politically constructed. But I’m still curious!
I don’t know if I will ever take a test. I ask myself: Should I be contributing to this system? Could I convince Sojourners to pay for my test if I were to write an article about it, thus shifting some of the ethical burden away from me as an individual?

News that scientists had for the first time recovered stem cells from cloned human embryos prompted dire warnings from religious leaders who say the research crosses a moral red line and could lead to designer babies.
Boston Cardinal Sean O’Malley, point man for the U.S. Catholic bishops on bioethical issues, said Wednesday that “this means of making embryos for research will be taken up by those who want to produce cloned children as ‘copies’ of other people.”
Human cloning “treats human beings as products,” O’Malley said on behalf of the bishops, “manufactured to order to suit other people’s wishes. … A technical advance in human cloning is not progress for humanity but its opposite.”
I AM AGAINST the death penalty in principle. The deliberate killing of prisoners does not demonstrate our society's respect for life, which we are trying to teach—especially to those who violate it. We simply should not kill to show we are against killing. It's also easy to make a, yes, fatal mistake, as alarming DNA testing has demonstrated, revealing some death row inmates to be innocent. In addition, the death penalty is clearly biased against poorer people, who cannot afford adequate legal representation, and is outrageously disproportionate along racial lines. The facts are that few white-collar killers sit on death row, and fewer are ever executed. And there is no evidence that capital punishment deters murder; the data just doesn't show that.
At a retreat I attended a couple of years ago, conservative activist Richard Viguerie approached me and said, "Jim, let's do something together to really shake up politics." Viguerie had become a friend, so I asked him what that might be. "I am a Catholic," Viguerie said. "I am against the death penalty, and I think it's time for conservatives and liberals who agree on that to begin to work together." I was fascinated at the thought of unlikely partners helping to accomplish that together. So we have had several dinner meetings over the last two years with both conservative and liberal leaders—mostly people of faith—to discuss the issue.
Here are some basic facts. There have been 1,312 executions since 1976, when the death penalty was reinstated following a 10-year moratorium. There were 43 prisoners killed in 2011, and 35 so far in 2012. As of April 2012, there were 3,170 people on death row. Forty-two percent are black, 43 percent are white, and 12 percent are Latino. Thirty-three states have the death penalty; 17 have abolished it and several have abolition legislation pending. Since 1973, 141 people have been exonerated and set free from death sentences because of new evidence—people who shouldn't have even been prisoners and were almost killed by the state due to false or faulty evidence. Eighteen of them were released because of DNA evidence. Who knows how many people have been executed unjustly?
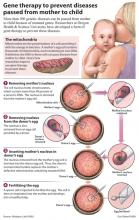
PORTLAND, Ore. – Future generations could be stripped of mutations such as hereditary blindness or maternal diabetes, after a breakthrough study at Oregon Health & Science University.
But the new technique is also one short step from genetic design of future generations, said Marcy Darnovksy of the California-based Center for Genetics and Society. "These powerful new technologies have a whole bunch of wonderful and appropriate uses – and a number of ways they can be misused.
The researchers, led by OHSU biologist Shoukhrat Mitalipov, modified unfertilized eggs for the first time, a technique that offers great promise as well as ethical pitfalls. Such research is banned in many countries.
Three years ago, the Russian-born Mitalipov made headlines with experiments that created monkeys with genetic material from three parents. Now, his team has done it with human cells, setting the stage for possible experiments with humans.
The procedure dealt with what's called mitochondrial DNA, the small part of the cell that turns food into energy. Mitochondrial disorders can lead to neuropathy (a type of dementia) and nervous system disorders such as Leigh disease.
In a study published Wednesday in the journal Nature, the OHSU team described successfully transferring DNA from donor cells into other donor cells, fertilizing the eggs to create 13 tiny early embryos of roughly 100 cells each. These pre-embryos, called blastocysts, were converted to embryonic stem cells for future research.
Key to the technique: replacing the defective mitochondrial DNA with healthy genetic material from the egg of a second woman.